Background: Hypercoagulability is a common blood alteration in newly diagnosed chemotherapy naïve patients with multiple myeloma. Multiple myeloma (MM) figures among malignancies that significantly increase the risk of venous thromboembolism (VTE). The rate of VTE is higher at the time of diagnosis and during the first months following initiation of first line therapy; approximately 10% of newly diagnosed MM (NDMM) will develop a VTE. Despite adequate thromboprophylaxis as per guidelines, the risk of residual VTE is not eliminated and remains as high as 12%. The optimization of VTE prevention in patients with MM is an unmet need. The exact mechanism of the increased risk of VTE is not yet fully understood. The identification of the procoagulant potential of cancer cells, which is related principally to tissue factor (TF) expression, attracts particular interest.
Aim: We conducted a longitudinal prospective observational study, to explore the relationship of MM with cellular and plasma hypercoagulability as well as tissue factor positive microparticles, aiming to identify the most relevant biomarkers, which could be used in a Risk Assessment Model (RAM) for VTE in combination with clinical risk factors.
Methods: Newly diagnosed patients (182) with multiple myeloma (NDMM) were prospectively enrolled. Patients were followed up for 12 months and the primary end-point was symptomatic objectively confirmed VTE. Patients were risk stratified and received thromboprophylaxis (none, aspirin on low molecular weight heparin (LMWH) based on previously published recommendations by the IMWG and EMN. Prior to treatment initiation and thromboprophylaxis initiation baseline biomarkers were obtained: Thrombin generation (TGT) in citrated PPP was assessed with the Thrombogram-Thrombinoscope® assay using PPP-reagent® 5 pm TF by Diagnostica Stago. The levels of P-Selectin and heparanase in plasma were measured with ELISA Kit (Cusabio Biotech and R&D Systems respectively). The procoagulant phospholipids clotting time was measured with STA-Procoag-PPL®, Levels of Factor VIIa were measured by Staclot VIIa-rTF®, D-Dimers (DDi) by Liatest D-Di (Diagnostica Stago, France), and Tissue Factor activity (TFa) by specific clotting based home test. TF expression MPC-derived microparticles (MPC-dMPs) were detected using a Zymuphen MP-TF activity kit (Hyphen BioMed, Neuville sur Oise, France). These were compared against values in a population of healthy individuals (n=30).
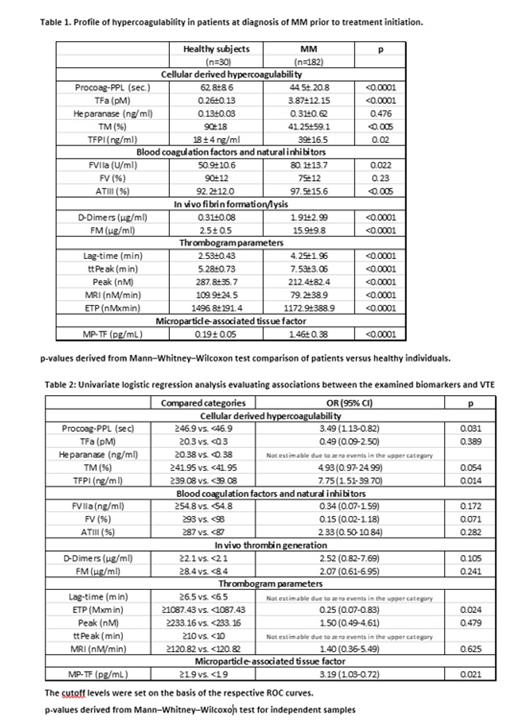
Results: The distribution of patients enrolled in the study is as follows: Median age was 67 years (36-86) and 52% of the population was male. Median time to follow up was 7 months (1-12 months). The control group (30 healthy individuals). The overall rate of symptomatic VTE during follow-up was 11.5% (n = 21 patients). Eleven out of 21 events (52%) occurred within 3 months from treatment initiation. Six of these patients did not receive any thromboprophylaxis; six patients were on aspirin at the time of the event and three were on LMWH.At inclusion, patients showed significantly increased levels of TFa, FVIIa, D-Dimers and FM, and significantly shorter Procoag-PPL® as compared to the group of healthy individuals. Levels of P-selectin and TM were significantly lower in patients as compared to healthy individuals. The levels of heparanase were not significantly different in the group of patients as compared to the healthy individuals. Overall thrombin generation was attenuated in patients compared to healthy individuals. Lag-time and ttPeak were significantly increased and Peak, MRI, and ETP were significantly lower as compared to the control group, MP-TF activity measured in NDMM significantly increased 1.46± 0.38 pg/mL compared to control subjects 0.19 ± 0.05 pg/mL (Table 1). Multivariate logistic regression analysis demonstrated that ETP, Procoag-PPL® and MP-TF were independently associated with VTE occurrence. (Table 2)
Conclusion: The prospective ROADMAP-CAT-MM study demonstrates the presence of pronounced cellular hypercoagulability in newly diagnosed chemotherapy naïve patients with MM characterized by decreased Procoag-PPL® clotting time, enhanced endothelial cell activation, and exhausted thrombin generation. The Procoag-PPL clotting time and the ETP and MP-TF were found to be independently associated with the risk of VTE and can be prospectively incorporated into a RAM for VTE in MM.
Disclosures
Kastritis:GSK: Honoraria, Research Funding; Pfizer: Honoraria, Research Funding; Janssen: Honoraria, Research Funding; Sanofi: Honoraria. Terpos:Takeda: Honoraria, Other: Travel expenses, Research Funding; Sanofi: Honoraria, Other: Travel expenses, Research Funding; Pfizer: Honoraria; Menarini/Stemline: Honoraria; Janssen: Honoraria, Research Funding; GSK: Honoraria, Research Funding; EUSA Pharma: Honoraria, Other: Travel expenses; ASTRA/Zeneca: Honoraria, Other: Travel Expenses; BMS: Honoraria; Amgen: Honoraria, Other: Travel Expenses, Research Funding.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal